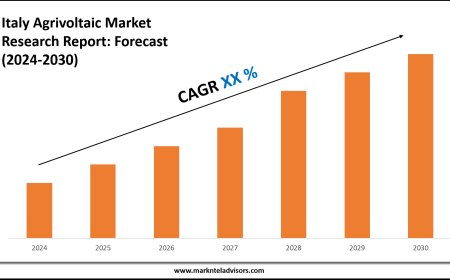
Section 321 Customs, FDA Prior Notice Web Entry Explained for Efficient Import Compliance
This web entry system collects essential product information to help the FDA monitor and inspect imported goods efficiently. It ensures that shipments meet safety standards and helps importers avoid delays or penalties.
Section 321 Customs of U.S. Customs law allows certain low-value shipments, typically those valued at $800 or less, to enter the country duty-free. However, this exemption does not remove the need for regulatory compliance. All food and FDA-regulated products entering under Section 321 still require an FDA Prior Notice submission through the FDA Prior Notice Web Entry system.This web entry system collects essential product information to help the FDA monitor and inspect imported goods efficiently. It ensures that shipments meet safety standards and helps importers avoid delays or penalties. Understanding the interplay between Section 321 and the Prior Notice requirement is crucial for food importers and businesses dealing with FDA-regulated items.
Understanding Section 321 Customs and FDA Prior Notice Web Entry
Section 321 allows certain shipments to enter the U.S. duty-free if they meet specific criteria, while the FDA Prior Notice system ensures compliance for food imports. Both are essential for importers managing low-value shipments, especially food items requiring prior notification before arrival.
Overview of Section 321 Exemptions
Section 321 exempts shipments valued at $800 or less from paying import duties and taxes. This de minimis threshold applies per person, per day, and applies only if the imported goods are not subject to other restrictions.These shipments bypass formal customs entry, accelerating processing and reducing paperwork. However, goods requiring oversight by Partner Government Agencies (PGAs) may need additional documentation even if under this threshold.The rule facilitates duty-free imports mainly for e-commerce and small businesses, allowing smoother access to U.S. consumers. Shipments exceeding $800 or under some PGA rules must follow standard entry procedures.
Legal Requirements for Section 321 Shipments
To qualify under Section 321, the total fair retail value of the shipment must not exceed $800. The shipment must be imported by one person on one day.Goods cannot violate other import restrictions, such as controlled substances or products restricted by specific agencies. The carrier or importer must provide accurate information about the shipment to customs.Section 321 benefits do not eliminate the need to comply with all other laws, such as safety, labeling, or quarantine regulations. Failure to meet these requirements may result in shipment denial or penalties.
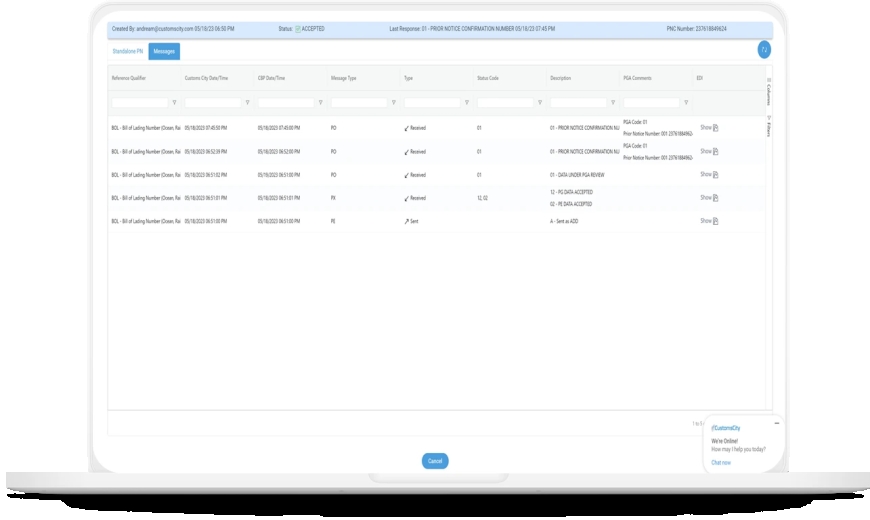
FDA Prior Notice Web Entry Process
The FDA mandates prior notice for all imported food products, regardless of value or quantity. This must be submitted through the FDA Prior Notice Web Portal before the shipment arrives in the U.S.The prior notice includes detailed shipment information, such as description, quantity, and importer details. For food shipments under Section 321, the HS tariff number must appear on the invoice.After submitting prior notice, importers receive a confirmation number, which must be given to the carrier for customs clearance. This preemptive communication helps the FDA review and inspect goods efficiently upon arrival.
Compliance and Best Practices for Importers
Importers must ensure precise filings and maintain thorough records to meet legal requirements under Section 321 and FDA Prior Notice regulations. Avoiding common errors reduces risk and supports smooth customs clearance for low-value shipments.
Filing Accurate Web Entries
Importers filing FDA Prior Notice must include all required data, such as HS tariff numbers and accurate product descriptions. TheFDA Prior Notice Web Portal is the official platform for submission.Entries under Section 321 require the shipments value not to exceed $800 per recipient per day. The prior notice number must be clearly provided to carriers for successful Customs and FDA clearance.Accurate and timely submission helps prevent delays and penalties. Double-check all shipment details, including the origin, importer information, and content specifics to ensure compliance.
Common Errors and How to Avoid Them
Frequent errors include missing tariff codes, incorrect shipment values, and late or no prior notice filings. These can result in shipment holds or fines.
To avoid issues, importers should:
- Verify HS codes against the products classification.
- Ensure the total value stays under the $800 threshold.
- File the FDA Prior Notice at leasttwo hoursbefore arrival for air, andfour hoursfor sea shipments.
Automated systems and compliance tools can reduce manual errors. Regular staff training on submission deadlines and documentation requirements is vital.
Recordkeeping and Documentation
Maintaining detailed records is critical for audits and compliance verification. Importers should keep copies of invoices, prior notice filings, and correspondence related to each shipment.
Records must include:
|
Document Type |
Retention Period |
Purpose |
|
Invoice |
At least 5 years |
Verify shipment value |
|
FDA Prior Notice |
At least 5 years |
Proof of submission |
|
Customs Entry Docs |
At least 5 years |
Compliance verification |
Consistent documentation supports dispute resolution and penalty mitigation if issues arise during Customs reviews.